(Syllabus) CBSE: National Eligibility Cum Entrance Test (NEET) "Chemistry"
Disclaimer: This website is NOT associated with CBSE, for official website of CBSE visit - www.cbse.gov.in

(Syllabus) CBSE: National Eligibility Cum Entrance Test (NEET) "Chemistry"
The Medical Council of India (MCI) recommended the following syllabus for National Eligibility-cum-Entrance Test for admission to MBBS/BDS courses across the country (NEET-UG) after review of various State syllabi as well as those prepared by CBSE, NCERT and COBSE. This is to establish a uniformity across the country keeping in view the relevance of different areas in Medical Education.
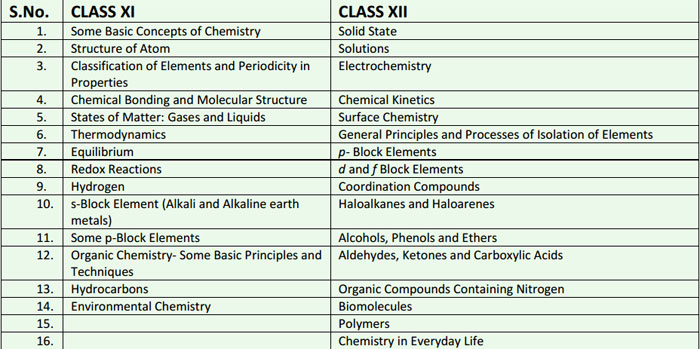
CONTENTS OF CLASS XI SYLLABUS
UNIT I: Some Basic Concepts of Chemistry
- General Introduction: Important and scope of chemistry.
- Laws of chemical combination, Dalton’s atomic theory: concept of elements, atoms and molecules.
- Atomic and molecular masses. Mole concept and molar mass; percentage composition and empirical and molecular formula; chemical reactions, stoichiometry and calculations based on stoichiometry.
UNIT II: Structure of Atom
-
Atomic number, isotopes and isobars. Concept of shells and subshells, dual nature of matter and light, de Broglie’s relationship, Heisenberg uncertainty principle, concept of orbital, quantum numbers, shapes of s,p and d orbitals, rules for filling electrons in orbitals- Aufbau principle, Pauli exclusion principles and Hund’s rule, electronic configuration of atoms, stability of half filled and completely filled orbitals.
UNIT III: Classification of Elements and Periodicity in Properties
- Modern periodic law and long form of periodic table, periodic trends in properties of elements- atomic radii, ionic radii, ionization enthalpy, electron gain enthalpy, electronegativity, valence.
UNIT IV: Chemical Bonding and Molecular Structure
- Valence electrons, ionic bond, covalent bond, bond parameters, Lewis structure, polar character of covalent bond, valence bond theory, resonance, geometry of molecules, VSEPR theory, concept of hybridization involving s, p and d orbitals and shapes of some simple molecules, molecular orbital theory of homonuclear diatomic molecules (qualitative idea only). Hydrogen bond.
UNITV: States of Matter: Gases and Liquids
-
Three states of matter, intermolecular interactions, types of bonding, melting and boiling points, role of gas laws of elucidating the concept of the molecule, Boyle’s law, Charle’s law, Gay Lussac’s law, Avogadro’s law, ideal behaviour of gases, empirical derivation of gas equation. Avogadro number, ideal gas equation. Kinetic energy and molecular speeds (elementary idea), deviation from ideal behaviour, liquefaction of gases, critical temperature.
- Liquid State- Vapour pressure, viscosity and surface tension (qualitative idea only, no mathematical derivations).
Click Here for Full Syllabus
<< Go Back to Main Page
Courtesy: CBSE